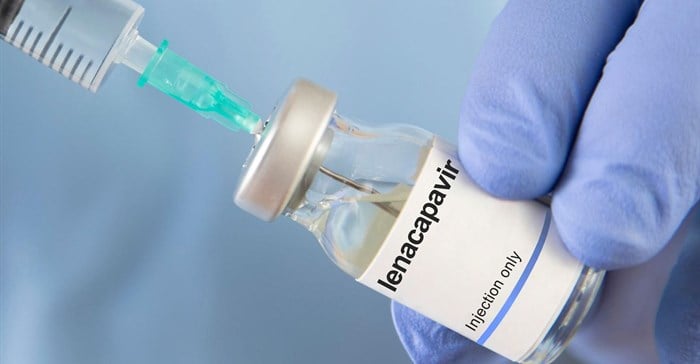
South Africa has made history as the first African nation to approve a twice-yearly HIV prevention injection, marking a major milestone in the country’s decades-long battle against HIV and AIDS.
The injectable drug, lenacapavir, will be introduced to the public from early 2026 following approval by the South African Health Products Regulatory Authority (SAHPRA).
The approval signals a new era for HIV prevention efforts in Africa’s most affected nation, where millions continue to live with the virus. Developed by US biopharmaceutical firm Gilead Sciences, lenacapavir was submitted for approval in March 2025 and has now been officially registered for use among HIV-negative adults and adolescents at high risk of infection.
During a national roundtable on lenacapavir access and sustainability, Health Minister Aaron Motsoaledi said the injection could revolutionise how South Africa tackles new infections.
“Lenacapavir represents a groundbreaking development in HIV prevention. Its long-acting formulation allows adults and adolescents to receive protection over six months with just two doses per year,” Motsoaledi said.
The South African National AIDS Council (SANAC), together with the Health Ministry, held discussions with key partners to evaluate infrastructure and funding strategies ahead of the rollout.
The injection will be given every six months after an initial under-the-skin dose, supported by two oral doses on the first and second days.
SAHPRA said the drug must be used alongside other protective measures such as condom use to reduce the risk of sexually transmitted infections.
“This product is the most effective HIV prevention measure thus far. The registration of lenacapavir is a game changer, given the high prevalence rate of HIV in South Africa,” SAHPRA CEO Boitumelo Semete Makokotlela said.
The approval process followed the European Medicines for All Procedure (EU-M4All), a mechanism that helps speed up access to essential medicines for non-European Union countries.
SAHPRA noted that this system enhances regulatory cooperation and helps countries strengthen access to lifesaving drugs faster.
To make the injection affordable, Gilead has partnered with six manufacturers — including one based in Egypt — to produce it at a cost of US$40 (Sh5,168) per person annually, down from the initial US$28,000 (Sh3.6 million).
The rollout will also benefit from a US$29.2 million (Sh3.7 billion) grant from the Global Fund to Fight HIV, TB, and Malaria, expected to cover 456,360 people over the next two years.
Partnerships with the Gates Foundation and local pharmaceutical companies are in progress to develop generic versions and eventually boost local production.
However, the Economic Freedom Fighters (EFF) have raised concerns over South Africa’s exclusion from Gilead’s licensing framework, describing it as “a modern form of pharmaceutical exploitation.”
“The registration of LEN is a step forward, but we must ensure local production to reduce costs and strengthen our healthcare system,” the party stated.
According to Minister in the Presidency Khumbudzo Ntshavheni, the first rollout will target 23 high-prevalence districts in six provinces, involving about 360 major public health facilities.
Experts estimate that between one and two million doses will be required annually to significantly curb new infections.
Currently, South Africa has about eight million people living with HIV, and roughly 1,000 adolescent girls and young women are infected each week.
If successfully implemented, lenacapavir could dramatically reduce HIV transmission rates and move South Africa closer to ending AIDS within the next two decades.